EQMS Software: Centralize Quality, Compliance & QMS Processes
Chakrapani KVC | 2 Jan 2026 | Not Modified
An eQMS (Electronic Quality Management System) is a Digital Platform designed to centralize, automate and streamline QMS processes within an Organization.
It serves as a comprehensive software solution that integrates all quality processes and compliance requirements across various departments, helping organizations manage workflows and documents more efficiently, improve compliance and enhance overall quality.
Additionally, an EQMS aids in controlling & improving Quality ensuring that products and services meet customer satisfaction and regulatory standards.
By transitioning from traditional paper-based systems to electronic solutions, eQMS streamlines quality management by digitizing documentation, automating workflows, and enhancing traceability and compliance. Electronic QMS enhances compliance, efficiency, and decision-making by automating workflows and minimizing manual errors.
Organizations implement a QMS to meet customer and regulatory requirements. An eQMS streamlines compliance with requirements like FDA 21 CFR Part 11, EU GMP Annex 11, ISO 13485, ISO 9001 etc. through predefined workflows, automated reminders, enhanced collaboration, and reporting.
What is the Difference Between an eQMS and a QMS?
EQMS = The strategy and system for quality management at an enterprise level (can be manual or digital).
eQMS = The electronic tool or software that enables digital execution of quality management processes.

The main difference between an eQMS and a QMS is that an eQMS is a digital, software-based solution accessed via computers, tablets, or mobile phones, enabling process automation and real-time collaboration.
In contrast, a traditional quality management system often depends on paper-based documentation and manual processes to manage quality management activities.
In the past, most QMS implementations relied on manual paperwork, physical records, and spreadsheets, making them prone to inefficiencies and errors. Physical documents and records can be scanned and converted into digital formats, such as PDFs, for electronic storage, creating a hybrid system that integrates both paper-based and electronic methods.
Compared to conventional systems, an eQMS provides automated workflow, secure data storage, remote access, and streamlined operations, reducing reliance on manual processes.
What is the Difference Between an Electronic QMS (eQMS) and Enterprise QMS (EQMS)?

The main difference between an electronic QMS (eQMS) and an Enterprise QMS (EQMS) is that eQMS solutions suit businesses of all sizes, and EQMS platforms address the complex needs of large organizations.
An electronic QMS digitizes and streamlines quality management processes such as document control, change control, training, audits, corrective actions, and others, providing efficiency and compliance benefits suitable for organizations of various sizes.
In contrast, an enterprise QMS offers extensive scalability, advanced integration capabilities across multiple departments or locations, robust analytics, and greater flexibility, specifically designed to handle the complex quality management requirements of large enterprises.
Depending on their needs, enterprises utilize one or more eQMS solutions. These solutions include specific functionalities within the eQMS, such as site functions, which enable organizations to restrict access to specific documents based on user roles assigned to individual sites.
EQMS (Enterprise Quality Management System)
Scope: Refers to a comprehensive, organization-wide quality management system that integrates processes, policies, and tools across multiple departments or business units.
Focus: Enterprise-level governance, compliance, and quality assurance.
Implementation: Can be manual, hybrid, or digital. It’s about the framework and strategy, not necessarily the technology.
Example: A global company implementing ISO 9001 processes across all sites, supported by standardized procedures and audits.
eQMS (Electronic Quality Management System)
Scope: Specifically refers to a software-based or digital platform for managing quality processes electronically.
Focus: Automation, digitization, and centralization of quality workflows (e.g., document control, CAPA, audits, training).
Implementation: Always technology-driven, typically cloud-based or on-premise software.
Example: Using platforms like MasterControl, Veeva, or Qualio to manage quality documentation and compliance digitally.
Difference between Paper Based QMS & eQMS
Paper-Based QMS
- Format: Physical documents, binders, printed SOPs, and manual records.
- Process: Manual handling of approvals, signatures, and updates.
- Accessibility: Limited to physical locations; hard to share globally.
- Compliance: Higher risk of errors, missing documents, and audit challenges.
- Change Management: Slow and prone to delays; version control is difficult.
- Cost: Lower initial cost but high long-term cost (storage, printing, labor).
- Audit Readiness: Time-consuming; requires manual collection and verification.
Electronic QMS (eQMS)
- Format: Digital platform/software for managing quality processes.
- Process: Automated workflows for approvals, CAPA, training, and audits.
- Accessibility: Cloud-based or on-premise; global access in real-time.
- Compliance: Easier to maintain regulatory compliance (ISO, FDA, HIPAA).
- Change Management: Fast, with automated version control and notifications.
- Cost: Higher initial investment but lower long-term operational cost.
- Audit Readiness: Instant access to records; improves efficiency and accuracy.
Key Reasons by Regulatory Bodies
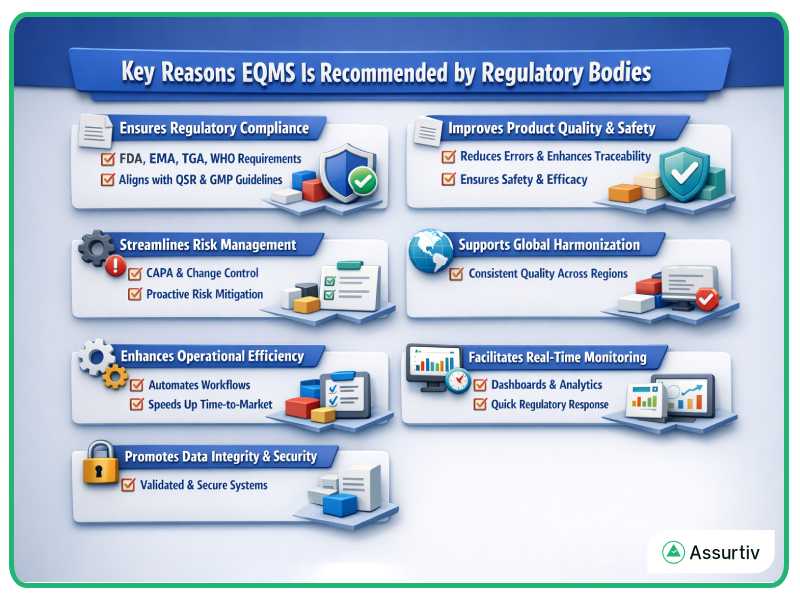
Ensures Regulatory Compliance
- 1. EQMS helps organizations meet stringent requirements set by FDA, EMA, TGA, and WHO by automating documentation, audits, and corrective actions.
- It aligns with frameworks like FDA’s Quality System Regulation (QSR) and EMA’s Good Manufacturing Practice (GMP) guidelines.
Improves Product Quality and Safety
- Centralized control over quality processes ensures that products meet safety and efficacy standards.
- Reduces human error and enhances traceability across the product lifecycle.
Streamlines Risk Management
- EQMS enables proactive identification and mitigation of risks through integrated CAPA (Corrective and Preventive Actions) and change control modules.
Supports Global Harmonization
- Facilitates consistent quality practices across international operations, which is essential for global regulatory approval.
- Aligns with WHO’s push for harmonized quality systems in pharmaceutical and medical device manufacturing.
Enhances Operational Efficiency
- Automates workflows such as training, document control, and audit management.
- Reduces manual paperwork and accelerates time-to-market for new products.
Facilitates Real-Time Monitoring and Reporting
- Provides dashboards and analytics for continuous quality improvement.
- Enables faster response to regulatory inspections and audits.
Promotes Data Integrity and Security
- Ensure secure, validated systems for managing sensitive quality data.
- Complies with data integrity standards like FDA’s 21 CFR Part 11 and EMA’s Annex 11.
Common Benefits Highlighted by All Authorities for the Usage of eQMS
- Improved Compliance: Automates for regulatory documentation and audit trails.
- Enhanced Data Integrity: Secures electronic records and ensures traceability.
- Streamlined CAPA and Change Control: Facilitates faster resolution of quality issues.
- Real-Time Monitoring: Enables dashboards and analytics for proactive quality management.
- Global Harmonization: Supports consistent quality practices across international sites.
An eQMS is no longer optional for regulated organizations. By digitizing quality processes, automating compliance, and enabling real-time collaboration, eQMS empowers businesses to reduce risk, improve efficiency, and consistently meet regulatory and customer expectations.



